Validation of an anti-α-Gal IgE fluoroenzyme-immunoassay for the screening of patients at risk of severe anaphylaxis to cetuximab, BMC Cancer
Por um escritor misterioso
Last updated 22 setembro 2024
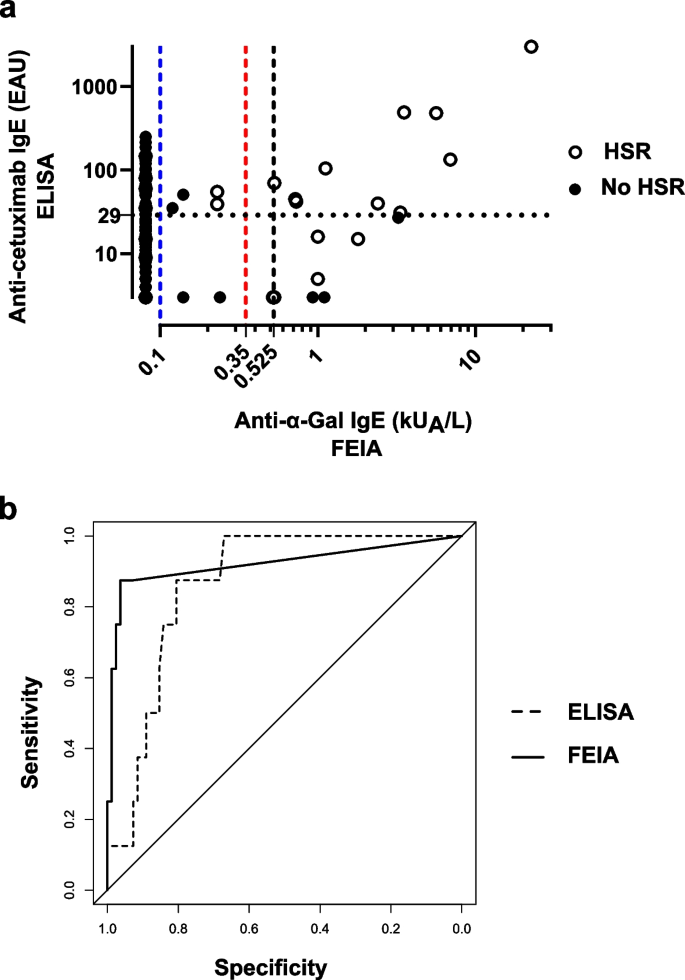
Background The link between immediate hypersensitivity reactions (HSR) following the first cetuximab infusion and the IgE sensitization against anti-galactose-α-1,3-galactose (α-Gal) is now well-established. An automated Fluoroenzyme-Immunoassay (FEIA) is available and may facilitate the screening of patients with anti-α-Gal IgE before treatment. Methods This study aimed to evaluate its performances as compared to a previously validated anti-cetuximab IgE ELISA, using 185 samples from two previously studied cohorts. Results Despite 21.1% of discrepancies between the two techniques, FEIA discriminated better positive patients and similarly negative ones with a ≥ 0.525 kUA/L threshold. Sensitivity was 87.5% for both tests, specificity was better for FEIA (96.3% vs ELISA: 82.1%). FEIA had a higher positive likelihood ratio (23.9 vs ELISA: 4.89) and a similar negative likelihood ratio (0.13 vs ELISA: 0.15). In our population, the risk of severe HSR following a positive test was higher with FEIA (56.7% vs ELISA: 19.6%) and similar following a negative test (0.7% vs ELISA: 0.8%). Conclusion Although the predictive value of the IgE screening before cetuximab infusion remains discussed, this automated commercial test can identify high-risk patients and is suitable for routine use in laboratories. It could help avoiding cetuximab-induced HSR by a systematic anti-α-Gal IgE screening before treatment.

PDF) Alpha-Gal-containing biologics and anaphylaxis

Description of the study population. HSR: Hypersensitivity reaction.
Frontiers Beyond IgE—When Do IgE-Crosslinking and Effector Cell Activation Lead to Clinical Anaphylaxis?

2022 White Paper on Recent Issues in Bioanalysis: FDA Draft Guidance on Immunogenicity Information in Prescription Drug Labeling, LNP & Viral Vectors Therapeutics/Vaccines Immunogenicity, Prolongation Effect, ADA Affinity, Risk-based Approaches, NGS, qPCR

(PDF) The role of IgE specific for galactose-α-1,3-galactose in predicting cetuximab induced hypersensitivity reaction: a systematic review and a diagnostic meta-analysis

Nationwide pharmacovigilance data for cetuximab-induced anaphylaxis and predictive model validation using prospective specific IgE detection - ScienceDirect

Prevalence of anti-cetuximab IgE. IgE levels were measured in serum

(PDF) Case Report About Fatal or Near-Fatal Hypersensitivity Reactions to Cetuximab: Anticetuximab IgE as a Valuable Screening Test
Frontiers Novel Approaches in the Inhibition of IgE-Induced Mast Cell Reactivity in Food Allergy

Mast cell activation test in the diagnosis of allergic disease and anaphylaxis - ScienceDirect

(PDF) Validation of an anti-α-Gal IgE fluoroenzyme-immunoassay for the screening of patients at risk of severe anaphylaxis to cetuximab

Manifestations of Antidrug Antibodies Response: Hypersensitivity and Infusion Reactions

Allergen Exposure: When Timing Is Everything - ScienceDirect

PDF) Alpha-Gal-containing biologics and anaphylaxis

The future of food allergy: Challenging existing paradigms of clinical practice - Anagnostou - 2023 - Allergy - Wiley Online Library
Recomendado para você
-
 TikTok viral: pide una torta de 'Elsa' de “Frozen” para el cumpleaños de su hija y el resultado fue decepcionante, Video, Video viral22 setembro 2024
TikTok viral: pide una torta de 'Elsa' de “Frozen” para el cumpleaños de su hija y el resultado fue decepcionante, Video, Video viral22 setembro 2024 -
 Puede fallar: una nena pidió su torta de Frozen, pero el resultado no fue el esperado22 setembro 2024
Puede fallar: una nena pidió su torta de Frozen, pero el resultado no fue el esperado22 setembro 2024 -
 elsa weird cake|TikTok Search22 setembro 2024
elsa weird cake|TikTok Search22 setembro 2024 -
 17 Bolos lindíssimos e apetitosos Fórum Outer Space - O maior fórum de games do Brasil22 setembro 2024
17 Bolos lindíssimos e apetitosos Fórum Outer Space - O maior fórum de games do Brasil22 setembro 2024 -
 Memetizando – Página: Array – Acabando com a sua produtividade – Blog de Humor – Tirinhas – Gifs – Prints Engraçados – Videos engraçados e memes do Brasil.22 setembro 2024
Memetizando – Página: Array – Acabando com a sua produtividade – Blog de Humor – Tirinhas – Gifs – Prints Engraçados – Videos engraçados e memes do Brasil.22 setembro 2024 -
 Seamens strike hi-res stock photography and images - Alamy22 setembro 2024
Seamens strike hi-res stock photography and images - Alamy22 setembro 2024 -
 UglyDolls (2019) - IMDb22 setembro 2024
UglyDolls (2019) - IMDb22 setembro 2024 -
 VIDEO: Niña se hace viral al ver su horrible pastel de cumpleaños de Frozen22 setembro 2024
VIDEO: Niña se hace viral al ver su horrible pastel de cumpleaños de Frozen22 setembro 2024 -
 Disney's Frozen Elsa Figure22 setembro 2024
Disney's Frozen Elsa Figure22 setembro 2024 -
Leda Miguel (@ledaemmontreal) • Instagram photos and videos22 setembro 2024
você pode gostar
-
 gif avatars? Stardew Valley Forums22 setembro 2024
gif avatars? Stardew Valley Forums22 setembro 2024 -
PC ] PES 2011 Offline PC Game ( Digital Download )22 setembro 2024
-
 The D.I.C.E. Awards 2022 categories & nominees22 setembro 2024
The D.I.C.E. Awards 2022 categories & nominees22 setembro 2024 -
 Camiseta John John Shadow Masculina - Alcateia Moda Masculina22 setembro 2024
Camiseta John John Shadow Masculina - Alcateia Moda Masculina22 setembro 2024 -
 AO ASHI T0122 setembro 2024
AO ASHI T0122 setembro 2024 -
 Boban Marjanovic spending the offseason in Jamaica22 setembro 2024
Boban Marjanovic spending the offseason in Jamaica22 setembro 2024 -
 🪴 WORLD END HAREM FANTASIA22 setembro 2024
🪴 WORLD END HAREM FANTASIA22 setembro 2024 -
 Re: Zero Anime deverá pintar na TV brasileira - Geek Project22 setembro 2024
Re: Zero Anime deverá pintar na TV brasileira - Geek Project22 setembro 2024 -
 Shiny Spiritomb Pokemon Trade Go22 setembro 2024
Shiny Spiritomb Pokemon Trade Go22 setembro 2024 -
 Sonderfahrzeug-Simulator 2012 - Metacritic22 setembro 2024
Sonderfahrzeug-Simulator 2012 - Metacritic22 setembro 2024

![PC ] PES 2011 Offline PC Game ( Digital Download )](https://down-my.img.susercontent.com/file/56ce455e5dd6f895769676004fc9bdf5)